|
9/18/2023 0 Comments Calculate pi of peptide chain 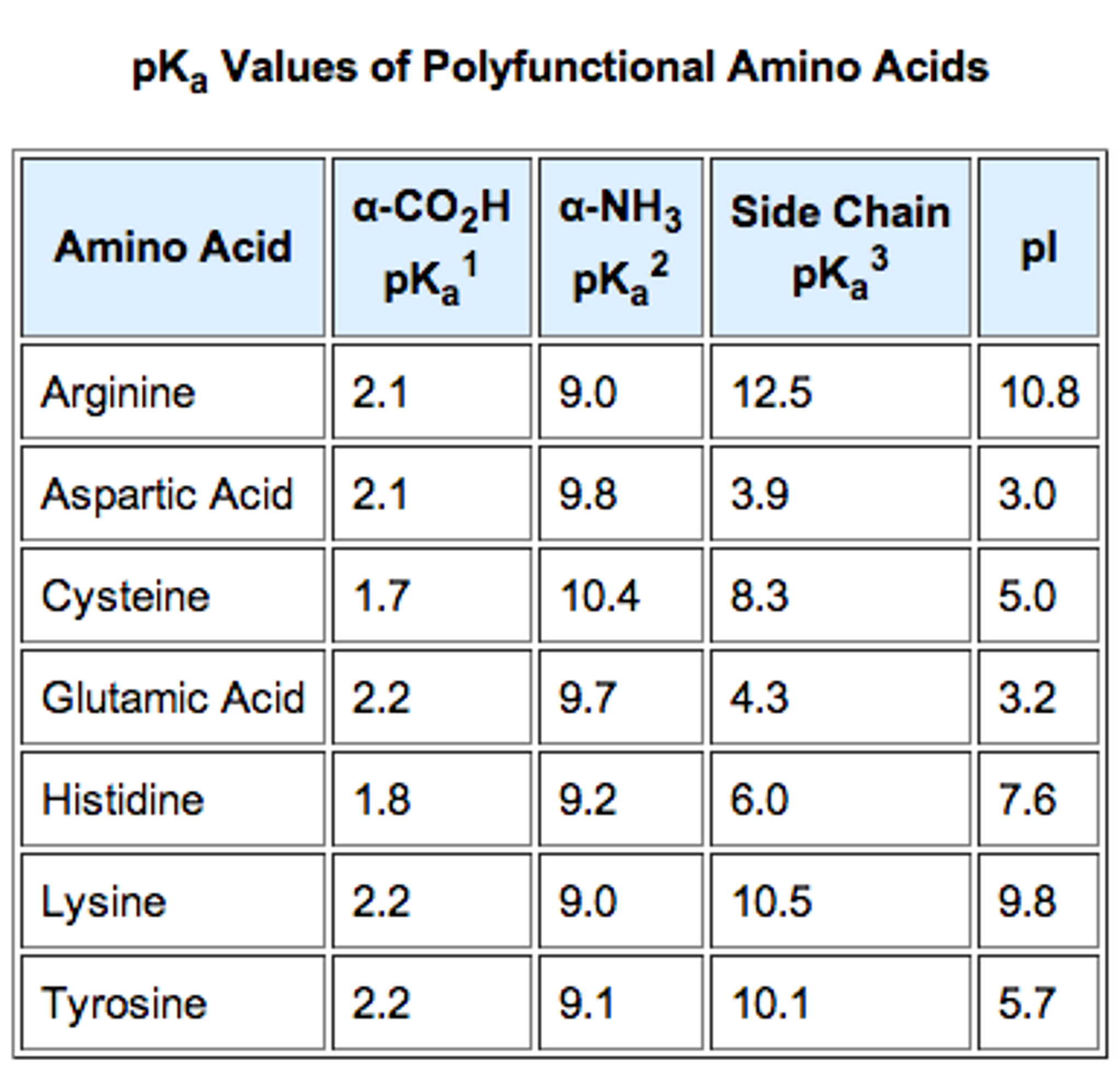
Each of these will have a name, a 3-letter abbreviation, and a 1 letter abbreviation. While there are hundreds of different amino acids that exist, there are only 20 that are used by the human body for protein synthesis. What do I need to know about Amino Acid Classification for the MCAT? Take the time to learn how to do this and make sure you understand it. An L-amino acid will not always be S, and a D-amino acid is not always R. Similarly, if the priority of the amino acid is NH2 > R > COOH, then an L-amino acid is the same as an S-amino acid and a D-amino acid is the same as a S-amino acid.Ī Common Misconception: L and D configurations of amino acids are different from R and S configurations of organic molecules. 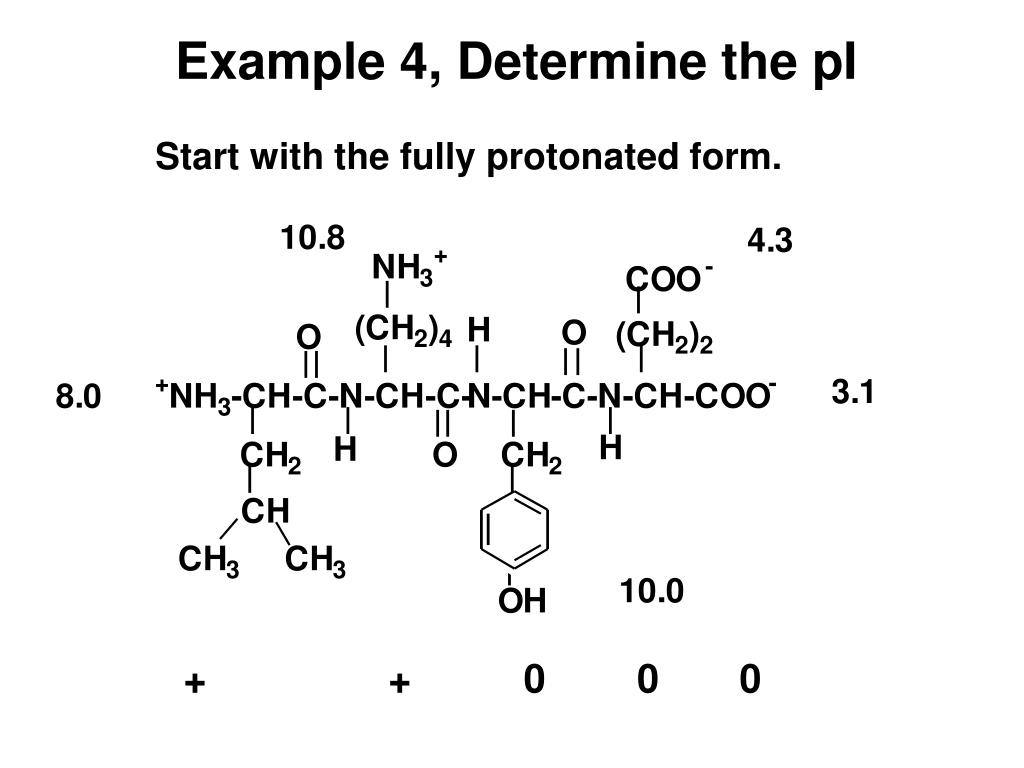
If the priority of the amino acid is NH2 > COOH > R, then an L-amino acid is the same as an S-amino acid and a D-amino acid is the same as a R-amino acid. To determine the configuration of an Amino Acid, you must compare the priority of the side groups of Amino Acids (A step-by-step instruction on how to do this will be covered in organic chemistry and determine the R- and S-configuration of carbon molecules). D-Amino Acids occur much less often in nature and, as a result, aren’t commonly used as a component in proteins. L-Amino Acids are more commonly found in nature, and in the human body, and are typically the type that are found in proteins. These configurations are commonly classified as L- or D-conformation. Although an amino acid may have the same functional components as another, the orientation of the side groups around the α carbon may change the overall properties of the molecule. When learning about Amino Acids for the MCAT, you also need to understand Amino Acid configurations to ensure you are maximising your potential of getting a good MCAT score. Different side chains will give the Amino Acid a different property, allowing it to interact with its environment in different ways. Arguably the most important component of the amino acid is the side chain. These groups are rotated around the α carbon in particular ways that allow them to interact with other amino acids. For acidic amino acids, the isoelectric point will be at lower pH as the acidic side chain will introduce an extra negative charge and for basic amino acids, the isoelectric point will be at higher pH as the basic side chain will introduce an extra positive charge.As you can see, there are 4 important components bound to a central carbon, called the α (alpha) carbon. For neutral amino acids, the side chains are neutral and the isoelectric point is given simply by the average of the pK a values of carboxylic acid and amine. Note: The isoelectric point is given by the average of the pK a values that involve the zwitterions, not just by the pK a values that describe the carboxylic acid group and the amine group. Since the isoelectric point is given by the average of the pK a values that involve the zwitterion, so we can write the formula for lysine as: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
